

First organicsClay minerals meet organic moleculesThere is no shortage of possible applications for the first organic molecules in clay mineral scenarios.Many of the possibilities were originally pointed out by A. G. Cairns Smith. The specific scenario described in the essay about the location of the origin of life suggests a particular likely role for the first organic molecules. That scenario places organisms in an environment where they are subjected to selection for large size, based on their mass and surface properties. A diagram illustrates the organisms:
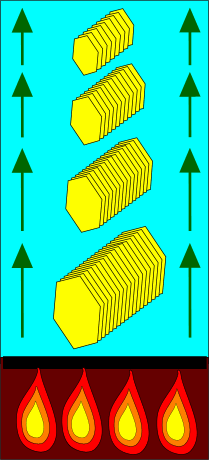 Larger organisms ride higher This diagram shows the way in which large organisms are stable low down, while small organisms are stable higher up.
Composite organismsThe specific proposal made here is that and the lowest organisms have another strategy available to them - besides increasing the their mass and cross-sectional area.If they could find a way to join with their neighbours, they could form composite organisms - which could be larger and heavier than any of the individual crystals that compose it. Organic molecules are in a good position to play the role of glue: if there is one thing that such molecules and are a good at, it is sticking things together.
Heavy aggregationsThe principle that aggregations of particles are often more likely to fall all out of suspension is a well-known one.Ionizers employ this principle to make dust particles stick together - so that their combined mass can overcome the convection currents that keep them airborne - and cause them and to a fall to the floor. The same principle is used by wine makers to cause impurities to adhere to one another and fall out of suspension. Clay particles are often used as the adhesive - due to their large surface area and otherwise benign nature.
DiagramThis diagram gives an indication of what a composite organism might look like:
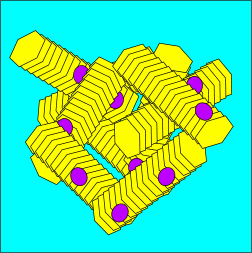 Heavy composite organism A composite organism like this would be large and heavy compared the individual crystals it is made up of. Each individual crystal adds to the mass of the whole - however the surface area increases more slowly than the mass - since individual crystals can act to shield each other from exposure to the surface. A simple experiment can be used to demonstrate the effect on a larger scale:
 Dropping paper clips into water The deeper the water you can drop the two bunches of paper clips into, the more obvious the effect is. I estimate the clump falls about 30% faster than a single paper clip - based on trials in a container of water which was about 80cm deep.
AdvantagesA composite organism might have the same growth rate as a monolithic core one of the same mass. However such a monolithic organism would be likely to have a larger cross-sectional area. It would consequently be much more difficult to to break.Since crystalline organisms reproduce by breaking, the composite organism would have a reproductive advantage - due to its individual crystals being easier to break and having a faster reproductive rate.
DisdvantagesUnlike their free-living relatives, composite organisms would face the issue of having their crystalline growth run into existing structures - potentially fusing crystals together. The chances of this happening are reduced if the crystals are relatively long and thin - and are increased if they are short and fat.This would be a problem for such organisms - but is probably not a show-stopping issue.
ReproductionComposite organisms would not themselves split in half - that would be unlikely. However, pieces of individual crystal might break off from around their edges.Such pieces would rise on the updraft. While many would rise to great heights, some would collide with other composite organisms living about them. These would contribute to the next generation of composite organisms.
Better glueOnce composite organisms form, there is selection to make better and stronger glues - in order to allow the composite organisms to better resist the updraught.Clay mineral crystals have an opportunity to control the types of particles that adhere to their surfaces. They can do this by changing the shape of the grooves that compose their surfaces.
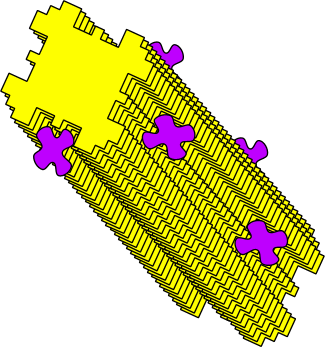 Catalytic grooves grab specified molecules
Future directionsEventually more complex relationships between the clay minerals and the organic molecules may form.Another essay discusses some of the possibilities in this area.
|